|
A project manager who begins a plan based on a complete and fully integrated template can help position senior management's expectations to minimize surprises.Ī 12-year development timeline is far too unwieldy for even the best manager to remember in detail. It is difficult to operate efficiently in an environment where there are no expectations.Įxecutives make better decisions when presented with realistic expectations. 1 Even with the creativity required within a function such as discovery, the department can benefit from having a rough guideline of how long it is allowed to experiment with a molecule before the company expects to have a lead compound to transition into nonclinical and CMC development. Behavioral studies show that productivity dramatically improves if there is some expectation of a deadline. "My work is too dependent on factors outside my control" is a common perspective. There are always those in an R&D organization who resist being held to a plan. It may even provide a means for controlling development costs. It creates a forum for project team member interaction and communication. It can provide a template that acts as an expert system for how things should be done. It can be a benchmark of company or team performance. The project plan may be used in many different roles. 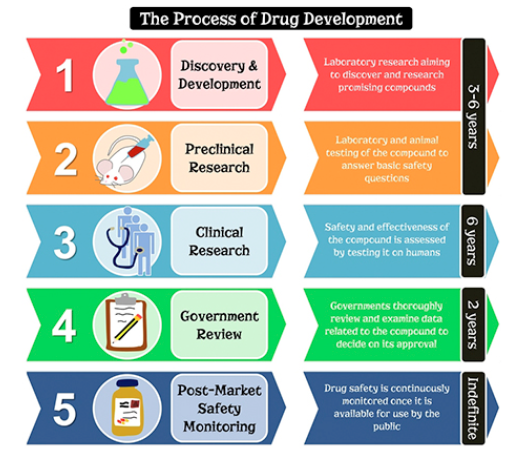
The project timeline self-adjusts based on the project's actual progress. Utilizing the Microsoft (MS) Project® software application, the template takes full advantage of the software's native functionality to properly organize, track, and present a complicated list of operational tasks and timelines. The estimated times used in the NCE project template are customized in case different assumptions need to be made by the project team, or if actual protocols become available with final estimates. The putative disease for this template is one that can be studied for efficacy in a 60-day "on-study" window in Phase II and III studies. Templates of those variants have also been developed however, for simplicity, only an NCE example is illustrated. From a project planning perspective, biologicals, new indications (sNDA), and generics (ANDA) have differences that would justify a modified template, particularly in the Nonclinical module and commonly in the CMC and Clinical modules. The NCE model assumes the compound to be a chemically synthesized IP. The 13 plan modules are: pharmacology discovery and efficacy proof of concept project initiation CMC development nonclinical development IND preparation clinical development CTD preparation 120-day safety update final labeling safety update FDA approval and launch preparation IND annual reports CTD periodic updates and product development and strategy. The modularity enables rapid customization for varying projects. The complete plan contains approximately 8400 tasks. It was built in 13 modular sections, utilizing the five functional domains of Discovery, CMC, Nonclinical, Clinical, and Regulatory. The NCE project template covers the broadest possible array of development milestones.  
The project template utilizes a regulatory backbone based on the IND and CTD tables of content. This article describes a fully integrated template of a project plan used to accelerate NDA-related activities, milestones, and deliverables for a New Chemical Entity (NCE) during an estimated 12-year development cycle. A project plan should be visible to the entire company as soon as discovery has a viable compound/lead or when a licensing agreement is being considered for an external compound. A complete, detailed, and integrated project plan is the best place to begin when focusing the valuable energies of functional departments. Departmental leads or representatives who participate in the project team negotiate, but they eventually accept the coach's prioritization and overall integration of each departmental plan into one project plan.Ī competent coach starts with a plan. Good project managers are schedule coaches, and they understand how departmental plans integrate into the overall project plan. Resident departmental experts rarely have the decades of experience necessary to intrinsically know how their work product dovetails with the requirements of other functional areas. Entire careers may be spent on just a few drug development programs. Yet, that is what good project managers are supposed to do: magic.ĭrug development is generally a slow process. When considering the unique knowledge in each of the five major domains of drug development, one quickly realizes that it takes a magician to coordinate the independent functional activities into a coherent and usable timeline. The 12-year roadmap to drug regulatory approval.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
